Redefining Small Molecule Analytics: Bridging the Gap Between LCŌĆōMS/MS and Immunoassays
Small molecule compounds (SMCs) are essential biomarkers in clinical diagnostics and therapy monitoring. However, their precise, high-throughput and at the same time cost-efficient determination still represents a central challenge in laboratory medicine.
SMCs (molecular weight <1000 Da), including thyroid hormones, sex hormones, vitamins and, in some cases, drugs, are of central importance for clinical diagnostics and therapy monitoring [1]. Due to their small size, simple structure, and a single detection epitope, classical sandwich immunoassays are not applicable [2]. Therefore, routine diagnostics are mainly based on competitive immunoassays. These determine the analyte concentration via competitive mechanisms, but are susceptible to matrix effects, which often leads to limited sensitivity, reduced accuracy in the low concentration range and a limited measurement range.
Currently, LCŌĆōMS/MS and competitive immunoassays dominate SMC analysis. LC-MS/MS offers high specificity and allows trace analysis, but requires elaborate instrumentation, specialized personnel, and limited sample throughput. Competitive immunoassays, on the other hand, are automated and widely available, but often have deficits in analytical precision. The combination of the advantages of both approaches has long been a central goal.
Against this background, non-competitive immunoassays for small molecules have been intensively investigated [2,5]. In contrast to competitive methods, they enable direct detection of the analyte and thus improve sensitivity and reproducibility. A particularly innovative development is the Noncompetitive Anti-Immune Complex Assay (NACA) technology.
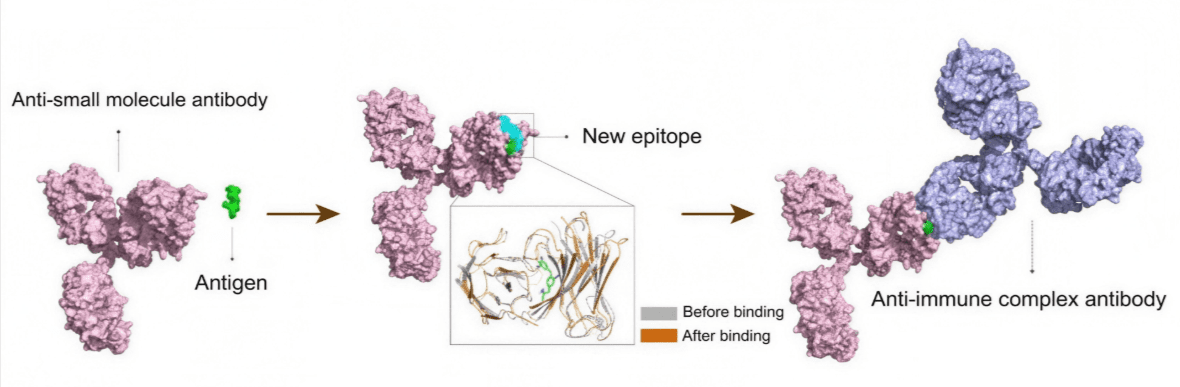
NACA technology uses secondary anti-immune complex antibodies that recognize newly formed epitopes that are created by the binding between the primary antibody and the target molecule [3,4]. This mechanism enhances the antigen-antibody interaction, increases binding stability and maximizes the efficiency of analyte binding, thereby significantly improving analytical accuracy.
As one of the early adopters, Snibe has successfully transferred this technology from research to clinical routine and established the Snibe NACA platform. This addresses the central weaknesses of conventional methods and offers five main advantages:
- High agreement with LCŌĆōMS/MS
- Wide linear measurement range for different clinical applications
- High sensitivity, especially in the low-concentration range
- High specificity with pronounced interference resistance
- High throughput with high cost efficiency at the same time
These features enable precise and scalable analysis of small molecules with minimal manual effort, balancing analytical performance and operational efficiency.
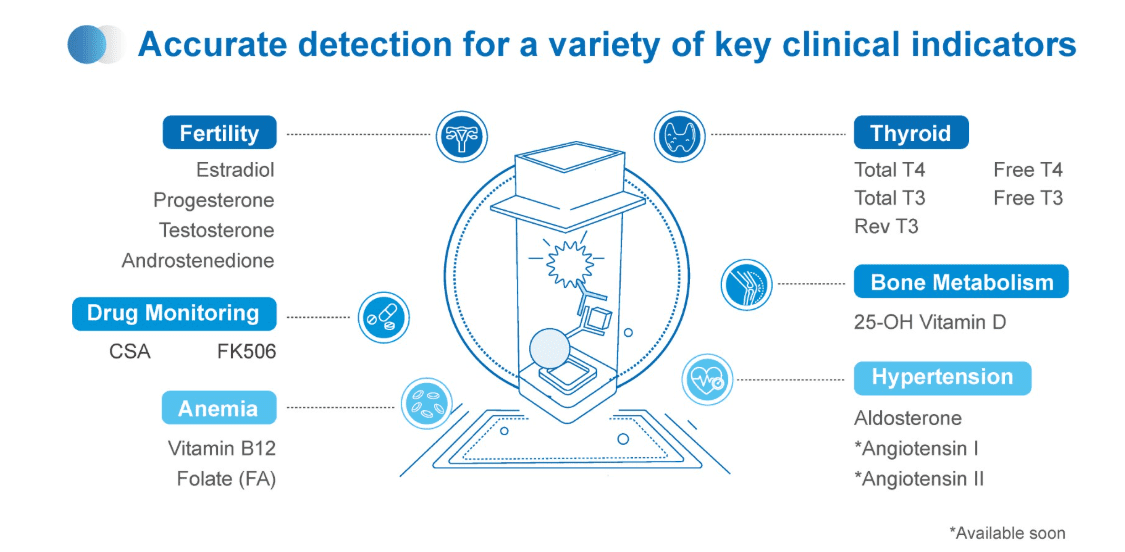
To date, NACA-based systems are used in over 100 countries and have generated more than 50 million test results. Applications span various specialties, including endocrinology, oncology, and reproductive medicine, and support early diagnosis and personalized therapeutic approaches.
Future developments aim to further increase sensitivity, expand the test portfolio and enable integration into digital health solutions. By closely interlocking technological innovation and clinical need, these advances are expected to both increase diagnostic precision and provide new insights into biological processes.
Reference:
[1] Li Y, et al. TrAC Trends in Analytical Chemistry, 2018, 103:198-208.
[2] Self C H, et al. Clinical chemistry, 1994, 40(11): 2035-2041.
[3] Nemazee D A, et al. Proceedings of the National Academy of Sciences, 1982, 79(12): 3828-3832.
[4] Voss Jr E W, et al. Molecular Immunology, 1988, 25(8): 751-759.
[5] Pulli T, et al. Analytical Chemistry, 2005, 77(8): 2637-2642.
For more information, visit https://www.snibe.com/en/
The respective companies are responsible for the content of the “Corporate News” section



