Nanobody penetrates cells and repairs defective CFTR channel in cystic fibrosis
A tiny antibody building block could fundamentally change the treatment of cystic fibrosis. Researchers at CharitÃĐ â UniversitÃĪtsmedizin Berlin and the Leibniz Research Institute for Molecular Pharmacology (FMP) have succeeded for the first time in developing a so-called nanobody that penetrates directly into human cells and stabilizes and repairs the most frequently mutated chloride channel (F508del-CFTR). The study was recently published in the journal Nature Chemical Biology.
The F508del mutation is present in about 90 percent of cystic fibrosis patients. It causes the CFTR channel to be folded incorrectly and degraded prematurely instead of working as a functioning chloride channel in the cell membrane. The result is thick mucus in the respiratory tract, chronic infections and a progressive loss of lung function.
The established triple therapy with elexacaftor, tezacaftor and ivacaftor (ETI) can raise CFTR function to about 50 percent of normal in many patients. The new nanobody binds specifically to the misfolded protein inside the cell, stabilizes it and supports correct folding. In cell cultures of cystic fibrosis patients, it increased channel activity to almost 90 percent of normal levels in combination with ETI.
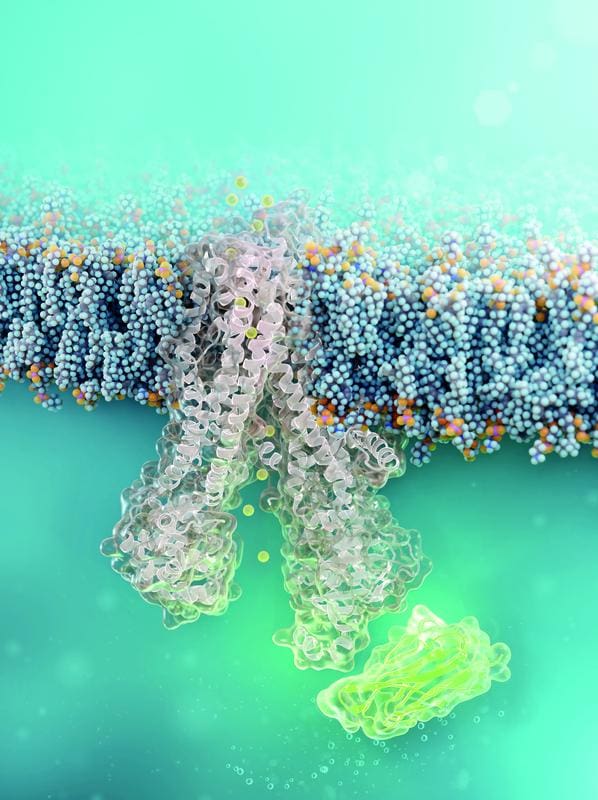
The researchers had chemically equipped the nanobody with cell-penetrating peptides that enable it to enter the cells. It remained bound to the mutated CFTR channel for at least 24 hours without damaging the cells. Functional tests showed a restored chloride transport function.
The approach is considered the first example of a functional cell-permeable antibody that is not only used to visualize or destroy cells, but also corrects disease-relevant protein misfolding directly inside the cell. The results suggest that with this strategy in combination with the existing CFTR modulators, a complete normalization of CFTR function may even be possible.
However, there are still considerable challenges to be overcome before clinical application â including a suitable inhaled formulation and the efficient penetration of the viscous mucus in the airways. The immune response to the nanobody also needs to be further investigated. The work was carried out within the framework of the Collaborative Research Centre 1449 “Dynamic Hydrogels at Biointerfaces”.
The researchers see potential in the concept not only for improved cystic fibrosis therapy, but also for other rare genetic diseases in which protein misfolding plays a central role.
Read Also:
Cystic Fibrosis Month of May: Focus on Care – MedLabPortal
Editor: X-Press JournalistenbÞro GbR
Gender Notice. The personal designations used in this text always refer equally to female, male and diverse persons. Double/triple naming and gendered designations are used for better readability. ected.