Chronic oxycodone exposure in pregnancy impairs fetal heart development
Chronic oxycodone exposure during pregnancy leads to profound changes in the proteome of placental small extracellular vesicles (PSEVs) and interferes with molecular signaling pathways associated with fetal cardiomyopathy. This is shown by a new preclinical study from the University of Nebraska Medical Center, which was published in the journal Extracellular Vesicles and Circulating Nucleic Acids .
The scientists led by Gurudutt Pendyala and Sowmya V. Yelamanchili investigated the effects of chronic maternal oxycodone treatment on the composition and function of placental EVs in an established rat model. These vesicles serve as important messenger carriers between mother and fetus and play a central role in fetal organ development.
Of 456 EV proteins identified, 107 were significantly dysregulated. Particularly striking was the significant downregulation of proteins such as Atp2a2 (calcium handling in the heart muscle), Lmna (nuclear stability), Tgfb3 (tissue remodeling), Agt (blood pressure regulation) and Sgce (membrane stability in the heart muscle). These changes affect central signaling pathways of hypertrophic and dilated cardiomyopathy. In addition, there were disturbances in energy metabolism (TCA cycle, fatty acid degradation), glycosylation and vesicular transport.
Die EVs aus oxycodon-exponierten Plazenten waren zudem kleiner und in h├Čherer Konzentration vorhanden, was auf ver├żnderte Biogenese oder Stress-induzierte Vesikulation hinweist.
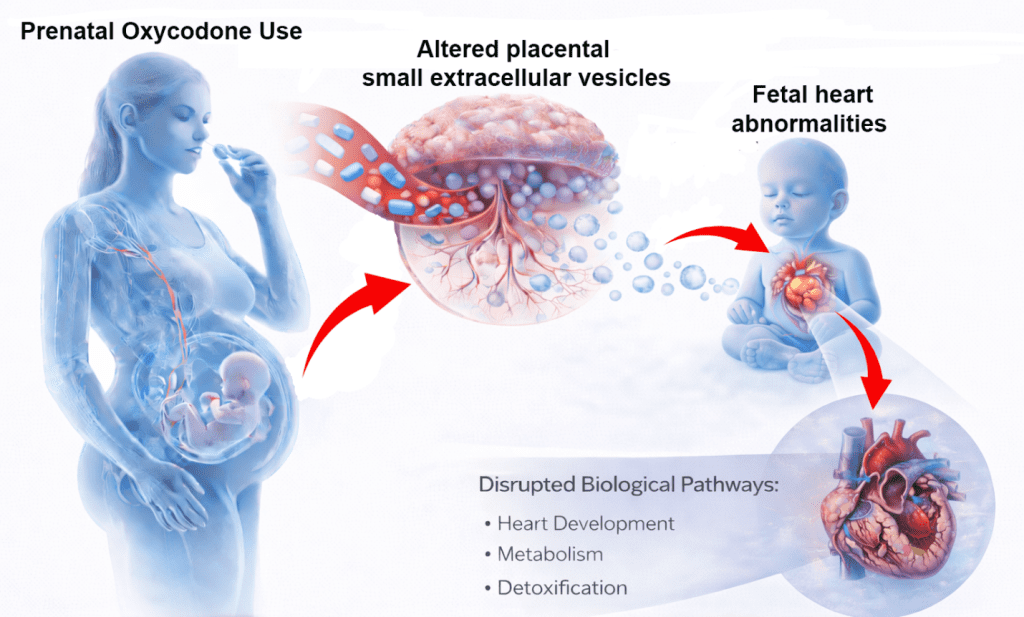
Opioids such as oxycodone are also widely used for pain management during pregnancy in the United States, but are leading to a growing number of newborns with withdrawal symptoms and long-term developmental risks. The placenta not only acts as a nutrient and oxygen supplier, but also as an active signaling transmitter via EVs. Disturbances in this system can have a lasting effect on fetal heart and vascular development.
The study provides for the first time a detailed proteomic characterization of placental EVs after chronic oxycodone exposure and links it to known cardiomyopathy pathways. The results are methodologically sound (combination of TEM, NTA, Western blot and label-free proteomics) and are well supported by bioinformatic analyses. The agreement with human MASH data in an earlier study by the same working group underlines the relevance of the model.
However, it is a preclinical rat model. Therefore, differences in placental structure, pharmacokinetics and fetal development between rats and humans must be considered. The sample size is moderate and independent validation of individual proteins (e.g. by Western blot or targeted assays) is still pending. Long-term studies on the actual heart function of the offspring are still lacking.
Nevertheless, the work provides important mechanistic insight and highlights the potential of placental EVs as biomarkers of opioid-related developmental risks. It lays the foundation for further translational research and possible EV-based diagnostics or interventions in perinatal medicine.
Original Paper:
Editor: X-Press Journalistenb├╝ro GbR
Gender Notice. The personal designations used in this text always refer equally to female, male and diverse persons. Double/triple naming and gendered designations are used for better readability. ected.